 
To form the Cs 2 + ion from Cs +, however, would require removing a 5 p electron from a filled inner shell, which calls for a great deal of energy: I 2 = 2234.4 kJ/mol for Cs. Q- If the formation of ionic lattices containing multiply charged ions is so energetically favorable, why does CsF contain Cs + and F − ions rather than Cs 2 + and F 2− ions?Ī- If we assume that U for a Cs 2 +F 2− salt would be approximately the same as U for BaO, the formation of a lattice containing Cs 2 + and F 2− ions would release 2291 kJ/mol (3048 kJ/mol − 756.9 kJ/mol) more energy than one containing Cs + and F − ions. Although the internuclear distances are not significantly different for BaO and CsF (275 and 300 pm, respectively), the larger ionic charges in BaO produce a much higher lattice energy. Lattice energies are directly proportional to the product of the charges on the ions and inversely proportional to the internuclear distance. If the first four terms in the Born–Haber cycle are all substantially more positive for BaO than for CsF, why does BaO even form?Ī- The answer is the formation of the ionic solid from the gaseous ions. Even though adding one electron to an oxygen atom is exothermic ( EA 1 = −141 kJ/mol), adding a second electron to an O −(g) ion is energetically unfavorable ( EA 2 = +744 kJ/mol)-so much so that the overall cost of forming O 2−(g) from O(g) is energetically prohibitive ( EA 1 + EA 2 = +603 kJ/mol). Q- Forming gaseous oxide (O 2−) ions is energetically unfavorable. The Born–Haber Cycle Illustrating the Enthalpy Changes Involved in the Formation of Solid Cesium Fluoride from Its Elements: Q- Arrange NaCl, MgS, AlN, and KBr in order of increasing lattice energy.Ī- The order of increasing lattice energy is KBr < NaCl < MgS < AlN. Q- Arrange GaP, BaS, CaO, and RbCl in order of increasing lattice energy.Ī- The order of increasing lattice energy is RbCl < BaS < CaO < GaP. 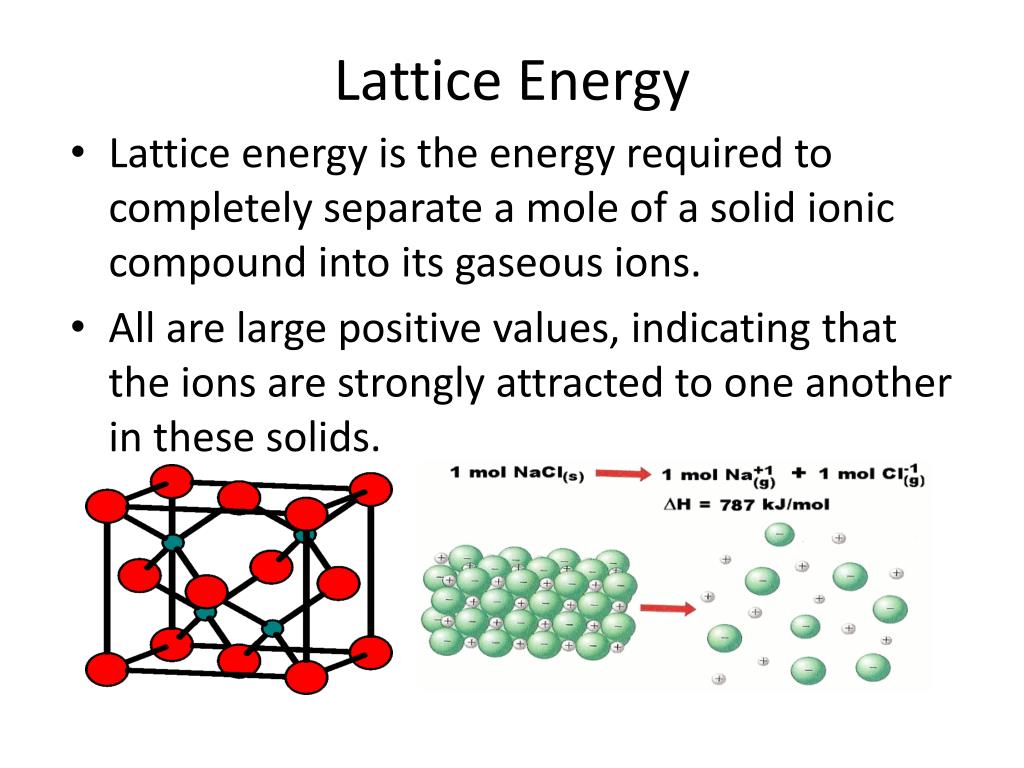
Lattice energy is the most important factor in determining the stability of an ionic compound. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
